Features old
Features old

Bespoke
Flexible, prompt and bespoke development.
Personalised homescreen to suit your role

Reliable
Cloud based system powered by AWS.
We have a dedicated support team ready to accommodate any changes or solve issues that may crop up over the course of your study.
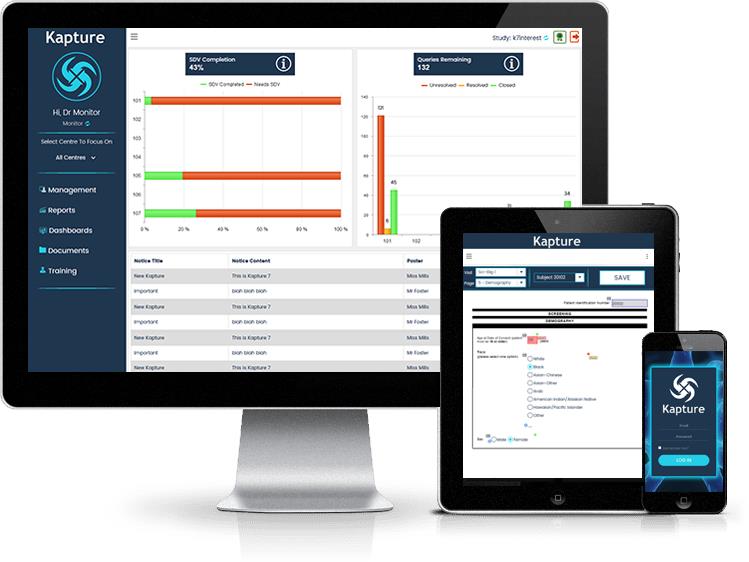
Responsive
Any Browser
Any Device (Tablet, Smartphone, Desktop/Laptop Computer)

Exporting and Integration
Charts, tables and reports export to PDF, Excel and images. This means you can integrate Kapture data collection and analytics with your exisiting documentation, presentation and communication systems.

Data security
Login controls
Versioning
Audit trails
Electronic signature
Robust and Compliant
21 CFR part 11 compliant.
Development follows strict protocols and all updates go through a rigourous testing procedure.

Customisation and Support
Tailored service, new features can be developed on customer request.
We release new versions specially for your study which enables us to adjust and configure Kapture to more closely match your needs.
Your trial, your Kapture
With all your Clinical trial system needs in one place, Kapture increases efficiency and reduces time to Clinical Study Reporting leading to shorter product submissions and thus saving money.
Individual clinical trials or whole development programmes can be quickly and flexibily deployed then managed either by your own Staff or by our own Team of experts.
Kapture is the only EDC solution which Investigators, Nurses, Patients, Data Managers and Project Managers alike love to use.
